Trust our science
Our clinical papers show how our products put you in control of your fertility journey, and maximize your chances of getting pregnant more quickly. That's because they are focussed on the accuracy of the two things you need to know to get pregnant - predicting and confirming the date of ovulation in each cycle.
What makes OvuSense different?
The patent protected OvuCore Sensor technology solves the issues caused by other methods by taking multiple overnight continuous Core Body Temperature (cCBT) measurements in the vagina using a thermistor with a resolution of 0.003 degrees Celsius - that means it's reading the differences to 1/1000th of a degree [a].
What are the benefits?
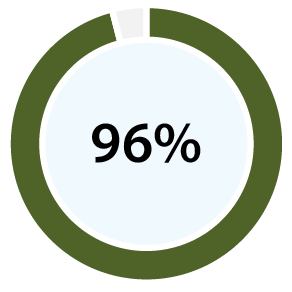
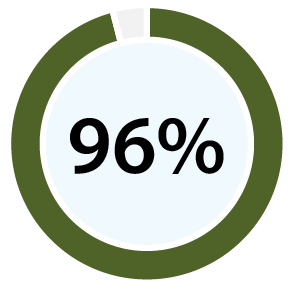
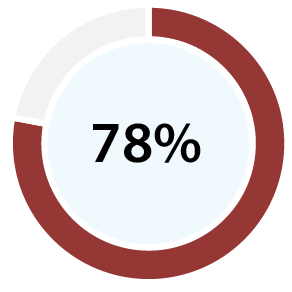
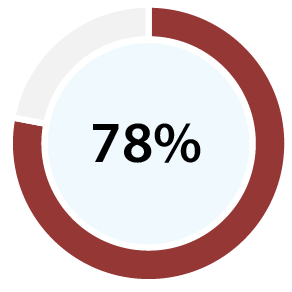
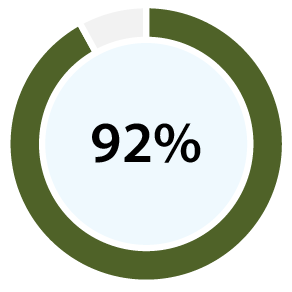
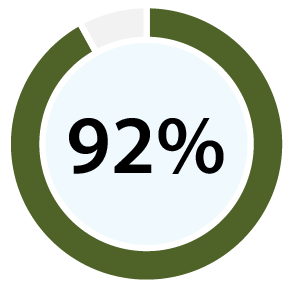
With 99% accuracy for confirming ovulation [b][d], and 96% positive predictive value for predicting ovulation [c][d], our clinical papers show OvuSense can save you up to 15 months trying to conceive compared with other devices. Take a look at the table at the bottom of this page to compare the accuracy of various other methods.
Up to 15 months quicker?
The 15 months difference is calculated as follows: According to clinical studies, your average chance of getting pregnant in any reproductive cycle is around 15%. [e] As a result 75% of the population will conceive within 5 months from the point at which they start out trying to conceive. A 100% accurate device would provide you with an exact understanding of the 8 day fertile window for each of those cycles and do so consistently.

With 99% accuracy, OvuSense provides 7-8 days of the window if your ovulation is regular, so if you're just starting out with regular ovulation timing it should take 5-6 months to get pregnant (100%/14-15%).
Other devices provide accuracies of up to 80% for the fertile window. In a 28 day cycle that means they can be up to 5 days out telling you on which days in your cycle you should try and conceive. With that wrong information your chances of getting pregnant can reduce to around 5% per cycle, and that means it could take up to 20 months to get pregnant (100%/5% =20) instead of 5-6.
What if I have irregular ovulation?
If you have irregular ovulation timing, then you probably already know it may take you longer to conceive. The really important fact is that urine strips and monitors (OPKs) and other temperature devices are much less likely to work for you. OvuSense is tracking the hormone released during ovulation and because it's able to predict that ovulation in real time during your cycle, it does work.
Urine strips annd monitors use hormones which don't behave as expected if you have an issue like Polycystic Ovarian Syndrome that causes variable ovulation, so you end up with false positive results or no positive results at all. [f] Other temperature devices use your result from a previous cycle to provide your fertile window so they simply don't work if your ovulation occurs on a different day in your next cycle. [g]
Added to the chances of getting pregnant more quickly with OvuSense, there's no messy urine strips, no confusing charts with multiple parameters, and no need to remember to test.
[a] The measuring resolution of a thermometer is the step between each temperature reading - a resolution of a normal 0.1 degree Celsius means a reading may be wrong by as much as 0.099 degrees Celsius, or in other words there are only 3 steps between a wrong and right ovulation result of 0.3 degrees Celsius rise, whereas 0.003 degrees resolution provides 1000 steps.
[b] The accuracy measures how many positive and absent ovulations the device confirms correctly.
[c] The positive predictive value measures how many of the real time predicted ovulations OvuSense gets right.
[d] The accuracy of ovulation confirmation by OvuSense is based on the data set originally published at the 2013 ASRM meeting as a quality index: Papaioannou S, Aslam M (2013), and developed in the key publication outlining the accuracy and positive predictive value of advance prediction of ovulation shown at the 2014 ESHRE meeting: Papaioannou S, Delkos D, Pardey J (2014).
[e] The chances of conceiving in each reproductive cycle are explored in the following clinical paper: Colombo B, Masarotto G (2000) Daily fecundability: first results from a new data base. Demogr Res 3: article 5. .
[f] The following papers together show that urine strips don't work if you have variable ovulation McGovern PG, Myers ER (2004); Irons DW, Singh M (1994); Lloyd R, Coulman CB (1989);
[g] The following papers together show that other temperature monitors and fertility bracelets don't work if you have variable ovulation Freundl G, Godehardt E (2003); Goodale BM, Shilaih M (2019)
Further clinical studies into our products have identified:
- the wide spread of day of ovulation in OvuSense user cycles:
Hurst BS, Citron C (2020) Normalised Ovulation Timing in Population using Core Body Temperature Monitoring to conceive S-194 Society for Reproductive Investigation, 2020 Annual Meeting.
- the association of atypical cycle patterns displayed by the OvuSense Pro product and ovulatory issues:
Karoshi M, Hurst B (2020) Atypical Temperature Patterns as an Aid to Identify Infertility Issues and Miscarriage Risk. P-628 European Society of Human Reproduction and Embryology 36th Annual Meeting.
- the atypical cycle patterns which led to the development of the OvuSense Pro product:
Hurst BS, Karoshi M (2020)
Atypical Core Body Temperature patterns and the wider implications for conditions related to pregnancy, infertility, and miscarriage risk
P-833
American Society for Reproductive Medicine 76th Annual conference. Poster; Fertility and Sterility - September 2020 Volume 114, Issue 3, Supplement, Pages e446–e447 |
Hurst BS, Pirrie A (2019) Atypical vaginal temperature patterns may identify subtle not yet recognised causes of infertility P-345 American Society for Reproductive Medicine 75th Annual conference.;, Fertility and Sterility - September 2019 Volume 112, Issue 3, Supplement, Pages e244–e245;
Our Patents
Our products are backed by a significant number of patents. Very few other products have this degree of proof of novel technology:
US Patents: US8684944, US8496597, US9155522, US9155523, US20190110692; European patents: EP1485690, EP2061380, DE20 2014 011 334, DE20 2014 011 335; Chinese patent: CN1650154
What is so important about clinical studies?
As you make a decision about which product to choose, clinical studies are one of the most important sources of information. However...
Not all studies are same. True clinical studies are done in controlled settings and reported as 'posters' at scientific meetings and in journals dedicated in our case to either Obstetrics and Gynecology, or by specialists in Reproductive Medicine. Studies presented this way must be “peer reviewed” by other specialists before they can be published. That's the way that medical professionals know they can trust the research.
As you consider other methods and products; take the time to check out the clinical studies that they quote. Some products don't provide any scientific backing, some talk about studies which are not reviewed by specialists, some are not published in specialist journals, and some don't provide you with useful information about how they can help you get pregnant.
Which fertility monitor is right for me?
Compare OvuSense with other methods for monitoring your ovulation and reproductive cycle.
As you consider which fertility monitor fits you best some typical things to consider are:
- How large a fertile window will it detect so you can maximize the time when you're trying to conceive
- How accurate is it at confirming ovulation - the less accurate the less chance of it helping you in your next cycle
- Can it predict ovulation using the current cycle data - something we like to call realtime prediction
- Does the system work for PCOS and irregular cycles
- Can it help your doctor screen for fertility issues
- Can it help you and your doctor monitor your medication or treatment
Each of the statistics quoted below are derived from clinical evidence available on fertility monitoring methods and products. Pro refers to the OvuSense Pro clinician portal. Click on the 'i' icon next to each method in the table, or directly on the method in the table on mobile devices to find out more about the clinical evidence.
OvuFirst (OF)
OvuSense (OS)
Fertility Tracker Apps (FTA)
Basal Body Temperature (BBT)
Skin Worn/ Fertility Bracelet Devices (SW/ FB)
Ovulation Predictor Kits and Monitors (OPK/ OPM)
Serum Progesterone (SP)
Ultrasound (US)
Fertility Testing Methods Key
- OF OvuFirst (Skin Temperature)
- OS Core OvuSense OvuCore (Core Body Temperature)
- BBT Basal Body Temperature
- FTA Fertility Tracker Apps
- SWD Skin Worn Devices
- FBD Fertility Bracelet Devices
- OPK Ovulation Predictor Kits (urine strips)
- OPM Ovulation Prediction Monitors (urine strips)
- SP Serum Progesterone
- US Ultrasound